(509) Elimination of Background Interference Using Spherotech and Commercial EDTA
.jpg)
Christine Peiter, CHS (ACHI) (she/her/hers)
– Lead Antibody Technologist, Gift of Life Michigan, United States
Speaker(s)
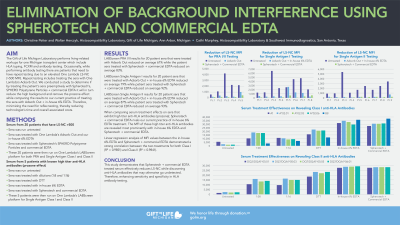
Aim: To compare serum treatments for reduction of high background using SPHERO™ Polystyrene Particles + Commercial EDTA (SP) for all antigen bead assays by Luminex LABScreen™ HLA antibody testing. This serum treatment process will be used to reduce the background for patients that exhibit elevated median fluorescent intensity (MFI) in the negative control (NC) and will remove the prozone effect without affecting sensitivity and specificity. This treatment minimizes the need for reflex testing, reduces technologist time and overall associated costs.
Methods: Five patient samples were tested to show that SP treatment removed the prozone effect as expected. Next, twenty-one patients with historically high NC >500 MFI were tested on One Lambda’s LABScreen platform for, PRA Class I/II and Single Antigen Class I/II. PRA Class I/II patient serum were run untreated, treated with Adsorb Out (AO) and, treated with SP. Single Antigen Class I/II patient serum were run untreated, treated with AO + Gift of Life Michigan’s (GOLM) 6% EDTA and, treated with SP. The results were compared for percent reduction of NC MFI and observed for any adverse effects. Additionally, twenty patient’s serum with HLA class I, class II or both classes of antibodies were tested by PRA Class I/II and Single Antigen Class I/II, untreated, GOLM 6% EDTA and, SP. Ensuring that both PRA I/II and Single Antigen I/II samples could have the same treatment applied before testing without adverse effects. The results were compared for accuracy, concordance, sensitivity, and specificity.
Results: The five patient samples exhibited prozone removal and was proven by linear regression analysis. The results confirm strong correlation between SP and GOLM 6% EDTA treated sera for both Class I (R2 = 0.9851) and Class II (R2 = 0.9841) single antigen beads. SP treatment effectively reduced non-specific complement activity without adverse effects. Furthermore, NC was reduced (Figure 1) and MFI-based analysis confirmed preservation of specificity and sensitivity (Figure 2). A Student's t-test showed no significant differences (p < 0.05) between GOLM and SP treated sera.
Conclusion: This study demonstrates that SP treated serum effectively reduces (NC) while revealing antibodies that may otherwise go undetected. Therefore, enhancing sensitivity and specificity in HLA antibody testing.
Methods: Five patient samples were tested to show that SP treatment removed the prozone effect as expected. Next, twenty-one patients with historically high NC >500 MFI were tested on One Lambda’s LABScreen platform for, PRA Class I/II and Single Antigen Class I/II. PRA Class I/II patient serum were run untreated, treated with Adsorb Out (AO) and, treated with SP. Single Antigen Class I/II patient serum were run untreated, treated with AO + Gift of Life Michigan’s (GOLM) 6% EDTA and, treated with SP. The results were compared for percent reduction of NC MFI and observed for any adverse effects. Additionally, twenty patient’s serum with HLA class I, class II or both classes of antibodies were tested by PRA Class I/II and Single Antigen Class I/II, untreated, GOLM 6% EDTA and, SP. Ensuring that both PRA I/II and Single Antigen I/II samples could have the same treatment applied before testing without adverse effects. The results were compared for accuracy, concordance, sensitivity, and specificity.
Results: The five patient samples exhibited prozone removal and was proven by linear regression analysis. The results confirm strong correlation between SP and GOLM 6% EDTA treated sera for both Class I (R2 = 0.9851) and Class II (R2 = 0.9841) single antigen beads. SP treatment effectively reduced non-specific complement activity without adverse effects. Furthermore, NC was reduced (Figure 1) and MFI-based analysis confirmed preservation of specificity and sensitivity (Figure 2). A Student's t-test showed no significant differences (p < 0.05) between GOLM and SP treated sera.
Conclusion: This study demonstrates that SP treated serum effectively reduces (NC) while revealing antibodies that may otherwise go undetected. Therefore, enhancing sensitivity and specificity in HLA antibody testing.
