(313) Teclistamab Causes Positive Flow Crossmatches

Xiaohai (Sam) Zhang, PhD
– director, Cedars Sinai Medical Center
Speaker(s)
Aim: Highly sensitized patients have a low likelihood of receiving a compatible donor offer. Various antibody therapies have been used to reduce antibody levels and increase the chances of a donor offer. However, these therapeutic antibodies can cause false positives in histocompatibility tests. Recently, our center began desensitizing transplant candidates using teclistamab, a bispecific antibody that binds both B-cell maturation antigen (BCMA) and CD3, recruiting and activating T cells to eliminate BCMA-expressing cells, such as plasma cells and plasmablasts. Here, we investigated whether teclistamab treatment causes positive complement-dependent cytotoxicity (CDC) and flow crossmatches.
Methods: One heart transplant candidate, previously desensitized with daratumumab, received five doses of teclistamab. Auto flow and CDC crossmatches with patient cells were performed using sera collected before and after treatment.
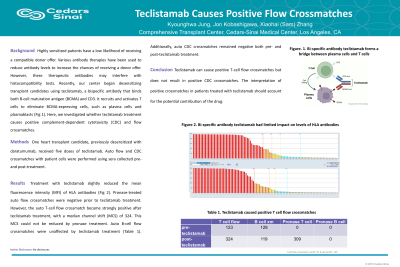
Results: Pronase-treated flow crossmatches were negative before teclistamab treatment. However, the auto T-cell flow crossmatch was strongly positive after teclistamab treatment, with a median channel shift (MCS) of 324. This MCS was not reduced by pronase treatment. Auto B-cell flow crossmatches were unaffected by teclistamab. Additionally, auto CDC crossmatches remained negative both before and after teclistamab treatment (Table 1).
Conclusion: Teclistamab can cause positive T-cell flow crossmatches but does not result in positive CDC crossmatches. The interpretation of positive crossmatches in patients treated with teclistamab should account for the potential contribution of teclistamab.
Methods: One heart transplant candidate, previously desensitized with daratumumab, received five doses of teclistamab. Auto flow and CDC crossmatches with patient cells were performed using sera collected before and after treatment.
Results: Pronase-treated flow crossmatches were negative before teclistamab treatment. However, the auto T-cell flow crossmatch was strongly positive after teclistamab treatment, with a median channel shift (MCS) of 324. This MCS was not reduced by pronase treatment. Auto B-cell flow crossmatches were unaffected by teclistamab. Additionally, auto CDC crossmatches remained negative both before and after teclistamab treatment (Table 1).
Conclusion: Teclistamab can cause positive T-cell flow crossmatches but does not result in positive CDC crossmatches. The interpretation of positive crossmatches in patients treated with teclistamab should account for the potential contribution of teclistamab.
