(303) Validation of a Bead-Based Anti-ABO-A Method Between Two Centres: One of These Things is a Lot Like the Other
- AH
Speaker(s)
Aim: Access to transplantation for ABO-O and -B candidates is reduced due to limited ABO-compatible donor pools. ABO-A2 kidneys have less A glycan expression than ABO-A1 thus ABO-O and -B candidates with low anti-A antibody levels may be eligible for ABO-A2 donors. Qualitative differences exist between A subtype glycans on ABO-A1 (A-II, -III, -IV) vsABO-A2 (A-II only) kidney and red blood cells (RBC) but ABO-A2-incompatible (ABO-A2i) eligibility is determined by anti-A antibody titres using A1 RBC. We developed a single antigen bead-based assay to measure subtype-specific ABO antibodies and used this test to compare to titre-eligibility to receive an ABO-A2-incompatible (ABO-A2i) kidney. This has been previously tested/published in a single centre cohort (Centre 1). Our aim was to test the potential MFI eligibility threshold for anti-A-II antibodies observed in Centre 1 to Centre 2.
Methods: A total of n=128 kidney transplant candidates in Centre 2 were tested/compared to n=141 candidates from Centre 1. Eligibility for ABO-A2i transplant was determined by A1 RBC titre (Table 1). All patient sera were re-tested retrospectively for IgG anti-A-II antibody by single antigen beads acquired on a FlexMap 3D system (no divider). Output shown as mean fluorescence intensity (MFI).
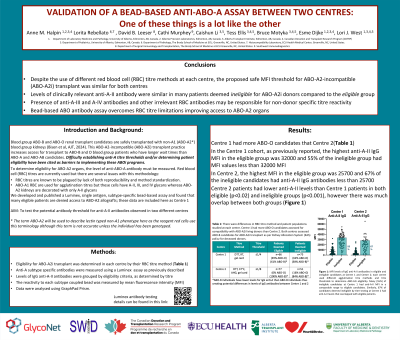
Results: As previously reported, in Centre 1 the highest anti-A-II IgG MFI in the eligible group was 32000; 55% of the ineligible group had MFI values <32000. In Centre 2, the highest MFI in the eligible group was 25700; MFI values were <25700 in 67% of the ineligible group. (Figure 1). The upper MFI range for the eligible patient group was similar for both centres. Anti-A-II antibody levels were significantly higher in both the eligible and ineligible groups in Centre 1 vs Centre 2 (p < 0.02 and p<0.001, respectively).
Conclusion: Despite each centre using different RBC titre methods and thresholds for ABO-A2i eligibility, the highest anti-A-II MFI for the eligible group was similar for both centres. Levels of clinically relevant anti-A-II antibody were similar in many patients deemed ineligible for ABO-A2i kidneys compared to the eligible group. This may be due in part to the contribution to anti-A titre by irrelevant anti-A-III and -IV antibodies. Our findings support that bead-based ABO antibody technologies can avoid unnecessarily restricted access to ABO-A2 organs created by limitations inherent to RBC titres.
Methods: A total of n=128 kidney transplant candidates in Centre 2 were tested/compared to n=141 candidates from Centre 1. Eligibility for ABO-A2i transplant was determined by A1 RBC titre (Table 1). All patient sera were re-tested retrospectively for IgG anti-A-II antibody by single antigen beads acquired on a FlexMap 3D system (no divider). Output shown as mean fluorescence intensity (MFI).
Results: As previously reported, in Centre 1 the highest anti-A-II IgG MFI in the eligible group was 32000; 55% of the ineligible group had MFI values <32000. In Centre 2, the highest MFI in the eligible group was 25700; MFI values were <25700 in 67% of the ineligible group. (Figure 1). The upper MFI range for the eligible patient group was similar for both centres. Anti-A-II antibody levels were significantly higher in both the eligible and ineligible groups in Centre 1 vs Centre 2 (p < 0.02 and p<0.001, respectively).
Conclusion: Despite each centre using different RBC titre methods and thresholds for ABO-A2i eligibility, the highest anti-A-II MFI for the eligible group was similar for both centres. Levels of clinically relevant anti-A-II antibody were similar in many patients deemed ineligible for ABO-A2i kidneys compared to the eligible group. This may be due in part to the contribution to anti-A titre by irrelevant anti-A-III and -IV antibodies. Our findings support that bead-based ABO antibody technologies can avoid unnecessarily restricted access to ABO-A2 organs created by limitations inherent to RBC titres.
