(302) Surrogate Crossmatches As a Tool to Define the True Incidence of HLA Class II de novo DSA

Luis G. Hidalgo, PhD, F(ACHI) (he/him/his)
– Director - Histocompatibility and Immunogenetics, University of Alabama - Birmingham, United States
Speaker(s)
Aim: De novo donor-specific HLA antibodies (dnDSA) targeting HLA-DQ and –DR antigens is a leading cause of graft loss. Misfolded HLA on SABs leads to artifactual reactivities that are clinically irrelevant and reporting such reactivities as DSA can change a patient’s clinical management. Physical crossmatches (XM) do not display misfolded HLA and can serve as a useful tool to verify potential DSAs. We confirmed possible dnDSAs against HLA-DR or HLA-DQ identified by SAB using flow cytometry surrogate B cell XM (surrBXM).
Methods: New reactivities against donor DR or DQ antigens that developed post-transplant were confirmed by performing surrBXM comparing previous to current sera. Leveraging our frozen cell stocks from nearly 6,000 donors, we identified cells homozygous for the specific HLA in question. dnDSA were defined as Negative in pre-transplant samples but increased reactivity (MFI≥1000) on SAB post-transplant. Conservative surrBXM reactivity categories, beyond the common binary XM output, were used to assess DSA reactivity: Negative - MCS ≤1 SD of control sera used to establish XM thresholds (≤33 MCS), Possible positive (1-2 SD; 34-66MCS), or Positive if >2 SD (≥67 MCS).
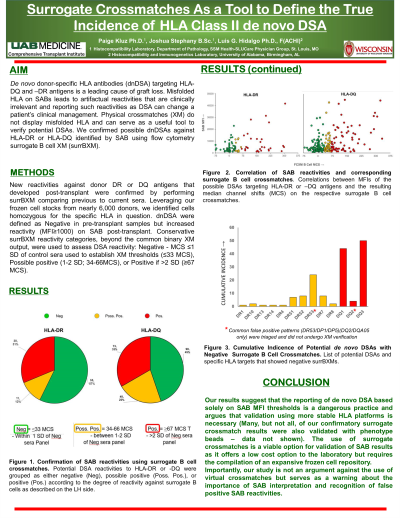
Results: 315 post-transplant samples with questionable DR or DQ reactivities were verified with surrBXM. For DR dnDSAs, 55/96 (57%) were negative on surrBXMs (Fig. 1A). For DQ, 98 of the 219 potential dnDSAs (45%) were also negative on surrBXMs. The high percent of DSAs negative on surrBXM was surprising given relatively high MFI values; DR (mean=4712, range=1088-22986), and DQ (mean=6059, range=1001-29954)(Fig. 1B). The specific HLA targets associated with negative surrBXMs showed high levels of disparity among HLA types Fig. 2. The majority, but not all, of specificities wth negative surrBXMs were also negative on Luminex phenotype beads.
Conclusion: Confirmation of potential dnDSAs using surrBXMs suggests that approximately half of dnDSAs are false positive if reported using basic MFI thresholds with SABs as the sole DSA testing method. Our data warns of the dangers of oversimplifying DSA testing and using SABs as the sole assay for DSA identification. Further, with the current push towards virtual XM, our study also demonstrates the necessity to not abandon cell-based testing given it’s potential utility as an additional platform to obtain accuracy in DSA reporting.
Footnotes: This abstract is a continuation of an ongoing study that will be presented as an oral presentation at the World Transplant Congress 2025.
Methods: New reactivities against donor DR or DQ antigens that developed post-transplant were confirmed by performing surrBXM comparing previous to current sera. Leveraging our frozen cell stocks from nearly 6,000 donors, we identified cells homozygous for the specific HLA in question. dnDSA were defined as Negative in pre-transplant samples but increased reactivity (MFI≥1000) on SAB post-transplant. Conservative surrBXM reactivity categories, beyond the common binary XM output, were used to assess DSA reactivity: Negative - MCS ≤1 SD of control sera used to establish XM thresholds (≤33 MCS), Possible positive (1-2 SD; 34-66MCS), or Positive if >2 SD (≥67 MCS).
Results: 315 post-transplant samples with questionable DR or DQ reactivities were verified with surrBXM. For DR dnDSAs, 55/96 (57%) were negative on surrBXMs (Fig. 1A). For DQ, 98 of the 219 potential dnDSAs (45%) were also negative on surrBXMs. The high percent of DSAs negative on surrBXM was surprising given relatively high MFI values; DR (mean=4712, range=1088-22986), and DQ (mean=6059, range=1001-29954)(Fig. 1B). The specific HLA targets associated with negative surrBXMs showed high levels of disparity among HLA types Fig. 2. The majority, but not all, of specificities wth negative surrBXMs were also negative on Luminex phenotype beads.
Conclusion: Confirmation of potential dnDSAs using surrBXMs suggests that approximately half of dnDSAs are false positive if reported using basic MFI thresholds with SABs as the sole DSA testing method. Our data warns of the dangers of oversimplifying DSA testing and using SABs as the sole assay for DSA identification. Further, with the current push towards virtual XM, our study also demonstrates the necessity to not abandon cell-based testing given it’s potential utility as an additional platform to obtain accuracy in DSA reporting.
Footnotes: This abstract is a continuation of an ongoing study that will be presented as an oral presentation at the World Transplant Congress 2025.
