(101) False-Positive Flow Cytometric Crossmatch in the presence of Therapeutic Monoclonal Antibodies Resolved by FlowDSA-XM Assay

Ping Rao, PhD
– Associate Director, City of Hope, United States
Speaker(s)
Aim: Therapeutic monoclonal antibodies (T-mAbs) are routinely used in treating patients with hematological malignancies. Some T-mAbs present in patient serum are reported to lead to false positive results when testing for HLA antibodies by flow cytometric crossmatch (FCXM). The FlowDSA-XM (FLDSA) assay uses beads to capture the HLA protein-antibody complex and is reported to discriminate between HLA and non-HLA antibodies. In this study, T-mAbs known to interfere with FCXM were tested at a range of concentrations to assess the impact on FCXM and whether these T-mAbs would interfere with the FLDSA assay.
Methods: Dilutions of intravenous immunoglobulin (IVIG), anti-thymocyte globulin (ATG), Rituximab (anti-CD20) and Teplizumab (anti-CD3) were created by the addition of the T-mAbs to a patient serum that had previously tested negative by both FCXM and by single antigen bead class I and II. This diluted T-mAb was added to healthy donor lymphocytes in a surrogate FCXM and a FLDSA (One Lambda) assay. The cells used in FCXM were stained for CD3 (T-cells), CD19 (B-cells) and anti-IgG. FLDSA was run according to the manufacturer’s protocol. FLDSA results are reported by three beads: one for HLA Class I antibodies, and two for HLA Class II antibodies. If either FLDSA class II bead is positive (IIa+ or IIb+), Class II is positive.
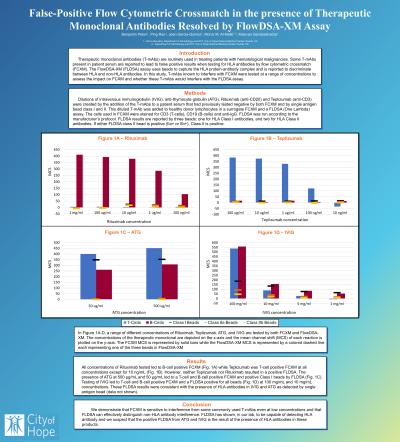
Results: All concentrations of Rituximab tested led to B-cell positive FCXM (Fig. 1A) while Teplizumab was T-cell positive FCXM at all concentrations except for 10 ng/mL (Fig. 1B). However, neither Teplizumab nor Rituximab resulted in a positive FLDSA. The presence of ATG at 500 µg/mL and 50 µg/mL led to a T-cell and B-cell positive FCXM and positive Class I beads by FLDSA (Fig. 1C). Testing of IVIG led to T-cell and B-cell positive FCXM and a FLDSA positive for all beads (Fig. 1D) at 100 mg/mL and 10 mg/mL concentrations. These FLDSA results were consistent with the presence of HLA antibodies in IVIG and ATG as detected by single antigen bead (data not shown).
Conclusion: We demonstrate that FCXM is sensitive to interference from some commonly used T-mAbs even at low concentrations and that FLDSA can effectively distinguish non-HLA antibody interference. FLDSA has shown, in our lab, to be capable of detecting HLA antibody and we suspect that the positive FLDSA from ATG and IVIG is the result of the presence of HLA antibodies in these products.
Methods: Dilutions of intravenous immunoglobulin (IVIG), anti-thymocyte globulin (ATG), Rituximab (anti-CD20) and Teplizumab (anti-CD3) were created by the addition of the T-mAbs to a patient serum that had previously tested negative by both FCXM and by single antigen bead class I and II. This diluted T-mAb was added to healthy donor lymphocytes in a surrogate FCXM and a FLDSA (One Lambda) assay. The cells used in FCXM were stained for CD3 (T-cells), CD19 (B-cells) and anti-IgG. FLDSA was run according to the manufacturer’s protocol. FLDSA results are reported by three beads: one for HLA Class I antibodies, and two for HLA Class II antibodies. If either FLDSA class II bead is positive (IIa+ or IIb+), Class II is positive.
Results: All concentrations of Rituximab tested led to B-cell positive FCXM (Fig. 1A) while Teplizumab was T-cell positive FCXM at all concentrations except for 10 ng/mL (Fig. 1B). However, neither Teplizumab nor Rituximab resulted in a positive FLDSA. The presence of ATG at 500 µg/mL and 50 µg/mL led to a T-cell and B-cell positive FCXM and positive Class I beads by FLDSA (Fig. 1C). Testing of IVIG led to T-cell and B-cell positive FCXM and a FLDSA positive for all beads (Fig. 1D) at 100 mg/mL and 10 mg/mL concentrations. These FLDSA results were consistent with the presence of HLA antibodies in IVIG and ATG as detected by single antigen bead (data not shown).
Conclusion: We demonstrate that FCXM is sensitive to interference from some commonly used T-mAbs even at low concentrations and that FLDSA can effectively distinguish non-HLA antibody interference. FLDSA has shown, in our lab, to be capable of detecting HLA antibody and we suspect that the positive FLDSA from ATG and IVIG is the result of the presence of HLA antibodies in these products.
