(117) A Robust AUC-Based Method for Eplet Calling in HLA Single Antigen Bead Assays: Reducing Variability and Enhancing Cohort-Level Profiling

Nicholas Borcherding, MD, PhD
– Assistant Professor, Washington University St Louis
Speaker(s)
Aim: Develop a robust methodology for eplet calling in SAB assays that reduces variability associated with single mean fluorescent intensity (MFI) cutoffs.
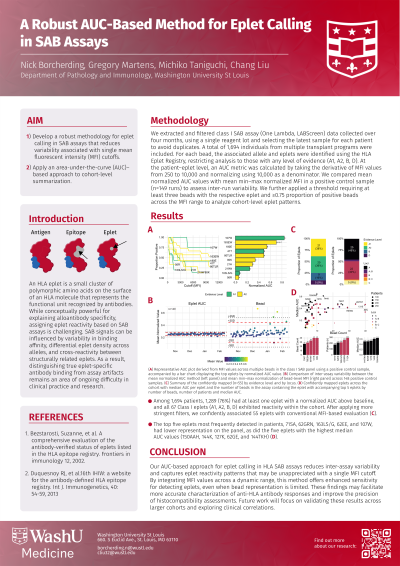
Methods: We extracted and filtered class I SAB assay data collected over four months, using a single reagent lot and selecting the latest sample for each patient to avoid duplicates. A total of 1,694 individuals from multiple transplant programs were included. For each bead, the associated allele and eplets were identified using the HLA Eplet Registry, restricting analysis to those with any level of evidence (A1, A2, B, D). At the patient–eplet level, an AUC metric was calculated by taking the derivative of MFI values from 250 to 10,000. We compared mean normalized AUC values with mean min–max normalized MFI in a positive control sample (n=148 runs) to assess inter-run variability. We further applied a threshold requiring at least three beads with the respective eplet and ≥0.75 proportion of positive beads across the MFI range to analyze cohort-level eplet patterns.
Results: Our AUC-based method successfully identified eplet patterns (A), demonstrating reduced variability compared to single-bead MFI measurements in the positive control sample (B). Among 1,694 patients, 1,289 (76%) had at least one eplet with a normalized AUC above baseline, and all 67 Class I eplets exhibited reactivity within the cohort. After applying more stringent filters, we confidently associated 55 eplets with conventional MFI-based evaluation (C). These eplets followed a power-law relationship when comparing their median AUC to the number of beads carrying each eplet (D). The top five eplets most frequently detected in patients—71SA, 62GRN, 163LS/G, 62EE, and 107W and top five eplets with the highest median AUC values (150AAH, 144K, 127K, 62GE, and 144TKH) had lower bead numbers.
Conclusion: In conclusion, our AUC-based approach for eplet calling in HLA SAB assays reduces inter-assay variability and captures eplet reactivity patterns that may be unappreciated with a single MFI cutoff. These findings may facilitate more accurate characterization of anti-HLA antibody responses and improve the precision of histocompatibility assessments. Future work will focus on validating these results across larger cohorts and exploring clinical correlations.
Methods: We extracted and filtered class I SAB assay data collected over four months, using a single reagent lot and selecting the latest sample for each patient to avoid duplicates. A total of 1,694 individuals from multiple transplant programs were included. For each bead, the associated allele and eplets were identified using the HLA Eplet Registry, restricting analysis to those with any level of evidence (A1, A2, B, D). At the patient–eplet level, an AUC metric was calculated by taking the derivative of MFI values from 250 to 10,000. We compared mean normalized AUC values with mean min–max normalized MFI in a positive control sample (n=148 runs) to assess inter-run variability. We further applied a threshold requiring at least three beads with the respective eplet and ≥0.75 proportion of positive beads across the MFI range to analyze cohort-level eplet patterns.
Results: Our AUC-based method successfully identified eplet patterns (A), demonstrating reduced variability compared to single-bead MFI measurements in the positive control sample (B). Among 1,694 patients, 1,289 (76%) had at least one eplet with a normalized AUC above baseline, and all 67 Class I eplets exhibited reactivity within the cohort. After applying more stringent filters, we confidently associated 55 eplets with conventional MFI-based evaluation (C). These eplets followed a power-law relationship when comparing their median AUC to the number of beads carrying each eplet (D). The top five eplets most frequently detected in patients—71SA, 62GRN, 163LS/G, 62EE, and 107W and top five eplets with the highest median AUC values (150AAH, 144K, 127K, 62GE, and 144TKH) had lower bead numbers.
Conclusion: In conclusion, our AUC-based approach for eplet calling in HLA SAB assays reduces inter-assay variability and captures eplet reactivity patterns that may be unappreciated with a single MFI cutoff. These findings may facilitate more accurate characterization of anti-HLA antibody responses and improve the precision of histocompatibility assessments. Future work will focus on validating these results across larger cohorts and exploring clinical correlations.
